Consistent supply and flexible distribution
- Dedicated PYLARIFY Customer Experience team for quick resolution of issues from order through delivery
- Multiple runs daily on weekends, based on location
- Coverage and reliability through an extensive, multi-partner 18F distributor network
Partners included, but not limited to:
- SOFIE
- PharmaLogic
- PETNET Solutions Inc,
A Siemens Healthineers Company - Jubilant Radiopharma
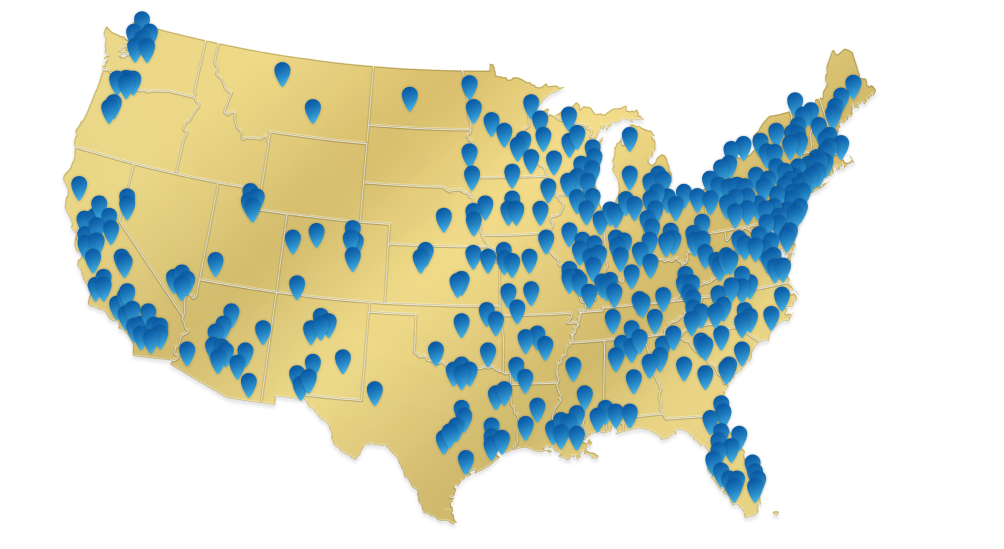
PYLARIFY is available nationwide1
- Over 1540 imaging centers
- Available in 48 states and Puerto Rico
Expanded release times and longer half-life to meet high patient volume and enable more flexible patient scheduling
PYLARIFY is available nationwide
For information about ordering PYLARIFY for your imaging site, and how to get started, contact PYLARIFY Customer Support at 1-800-964-0446 M-F 8:00 am-7:00 pm ET, or email cspyl@lantheus.com.

For information about ordering PYLARIFY for your imaging site, and how to get started, contact PYLARIFY Customer Support at 1-800-964-0446 M-F 8:00 am-7:00 pm ET, or email cspyl@lantheus.com.
PYLARIFY Imaging Site Locator
Please enter your ZIP code to locate the nearest imaging site that offers PYLARIFY®.
For information about ordering PYLARIFY for your imaging site, and how to get started, contact PYLARIFY Customer Support at 1-800-964-0446 1-800-964-0446 M-F 8:00 am-7:00 pm ET, or email cspyl@lantheus.com.
This may not be a comprehensive list. The sites listed are provided as an informational resource only.
Lantheus does not warrant, whether expressed or implied, that the Locator is accurate or complete. Lantheus does not recommend or endorse any site included in the Locator for any purpose. It is your responsibility to select a site and you assume full responsibility for your interactions with any site you contact through the Locator.
Lantheus is not responsible for the medical care or advice given by any site you choose to contact.
This free service provides a list of sites that may be qualified to provide PYLARIFY imaging. These sites do not pay a fee to be listed in the PYLARIFY Imaging Site Locator (“Locator”), and their participation does not impose any requirements on the manner in which they treat or provide services to patients. Participating sites are listed based on the geographical search information entered. Some of the participating sites may have other financial relationships with Lantheus as recipients of research grants or other similar payments.
Sites may request to be removed from the Locator and Lantheus retains the right to remove sites from the Locator.
To remove your facility from this list or request a specific change/update to a current site already in the Locator, contact us at pylcenters@lantheus.com.
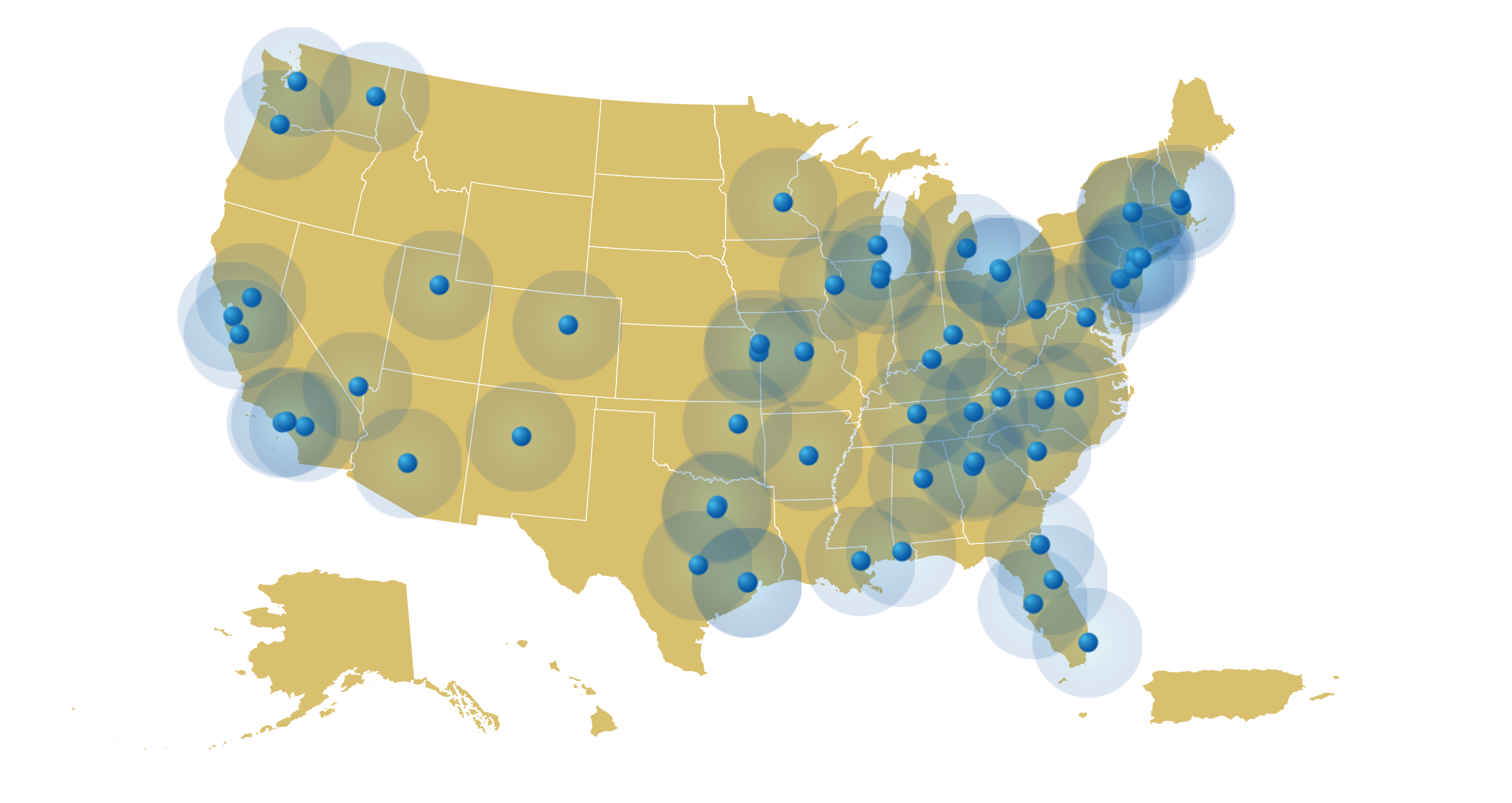
Available Now
- Alabama, Birmingham
- Arizona, Phoenix
- Arkansas, Little Rock
- California, Culver City
- California, Gilroy
- California, Loma Linda
- California, Sacramento
- California, San Francisco
- Colorado, Denver
- Florida, Fort Lauderdale
- Florida, Jacksonville
- Florida, Sanford
- Florida, Tampa
- Georgia, Atlanta
- Illinois, Chicago
- Illinois, Romeoville
- Iowa, Davenport
- Kansas, Kansas City
- Kentucky, Louisville
- Louisiana, Covington
- Massachusetts, Boston
- Massachusetts, Haverhill
- Michigan, Detroit
- Minnesota, Minneapolis
- Missouri, Columbia
- Missouri, Kansas City
- Nevada, Las Vegas
- New Jersey, Hackensack
- New Jersey, Somerset
- New Jersey, Totowa
- New Mexico, Albuquerque
- New York, Albany*
- New York, Bronx
- North Carolina, Raleigh
- North Carolina, Winston-Salem
- Ohio, Cincinnati
- Ohio, Cleveland
- Ohio, Oakwood Village
- Oklahoma, Tulsa
- Oregon, Portland
- Pennsylvania, Philadelphia
- Puerto Rico, San Juan
- South Carolina, Columbia
- Tennessee, Gray
- Tennessee, Knoxville
- Tennessee, Nashville
- Texas, Dallas*
- Texas, Houston*
- Utah, Salt Lake City
- Virginia, Sterling
- Washington, Seattle
- Washington, Spokane
- West Virginia, Morgantown
- Wisconsin, Milwaukee
- Kentucky, Louisville
- Louisiana, Covington
- Massachusetts, Boston
- Massachusetts, Haverhill
- Michigan, Detroit
- Minnesota, Minneapolis
- Missouri, Columbia
- Missouri, Kansas City
- Nevada, Las Vegas
- New Jersey, Hackensack
- New Jersey, Somerset
- New Jersey, Totowa
- New Mexico, Albuquerque
- New York, Albany*
- New York, Bronx
- North Carolina, Raleigh
- North Carolina, Winston-Salem
- Ohio, Cincinnati
- Ohio, Cleveland
- Ohio, Oakwood Village
- Oklahoma, Tulsa
- Oregon, Portland
- Pennsylvania, Philadelphia
- Puerto Rico, San Juan
- South Carolina, Columbia
- Tennessee, Gray
- Tennessee, Knoxville
- Tennessee, Nashville
- Texas, Dallas *
- Texas, Houston *
- Utah, Salt Lake City
- Virginia, Sterling
- Washington, Seattle
- Washington, Spokane
- West Virginia, Morgantown
- Wisconsin, Milwaukee
Available Now
Partners included, but not limited to:



- *2 manufacturing sites
- Trademarks, registered or otherwise, are the property of their respective owner(s).
- Use of this information is subject to the terms of our Legal Notice and Privacy Policy.
INDICATION
PYLARIFY (piflufolastat F 18) Injection is a radioactive diagnostic agent indicated for positron emission tomography (PET) of prostate-specific membrane antigen (PSMA) positive lesions in men with prostate cancer:
- with suspected metastasis who are candidates for initial definitive therapy.
- with suspected recurrence based on elevated serum prostate-specific antigen (PSA) level.
IMPORTANT SAFETY INFORMATION
Contraindications
None.
Warnings and Precautions
Risk of Image Misinterpretation
Imaging interpretation errors can occur with PYLARIFY imaging. A negative image does not rule out the presence of prostate cancer and a positive image does not confirm the presence of prostate cancer. The performance of PYLARIFY for imaging of patients with biochemical evidence of recurrence of prostate cancer seems to be affected by serum PSA levels. The performance of PYLARIFY for imaging of metastatic pelvic lymph nodes prior to initial definitive therapy seems to be affected by risk factors such as Gleason score and tumor stage. PYLARIFY uptake is not specific for prostate cancer and may occur with other types of cancer as well as non-malignant processes and in normal tissues. Clinical correlation, which may include histopathological evaluation of the suspected prostate cancer site, is recommended.
Hypersensitivity Reactions
Monitor patients for hypersensitivity reactions, particularly patients with a history of allergy to other drugs and foods. Reactions may be delayed. Always have trained staff and resuscitation equipment available.
Radiation Risks
Diagnostic radiopharmaceuticals, including PYLARIFY, expose patients to radiation. Radiation exposure is associated with a dose-dependent increased risk of cancer. Ensure safe handling and preparation procedures to protect patients and health care workers from unintentional radiation exposure. Advise patients to hydrate before and after administration and to void frequently after administration.
Adverse Reactions
The most frequently reported adverse reactions were headaches, dysgeusia and fatigue, occurring at rate of ≤2% during clinical studies with PYLARIFY. In addition, a delayed hypersensitivity reaction was reported in one patient (0.2%) with a history of allergic reactions.
Drug Interactions
Androgen deprivation therapy (ADT) and other therapies targeting the androgen pathway, such as androgen receptor antagonists, may result in changes in uptake of PYLARIFY in prostate cancer. The effect of these therapies on performance of PYLARIFY PET has not been established.
To report suspected adverse reactions for PYLARIFY, call 1-800-362-26681-800-362-2668 or contact FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
For important risk and use information about PYLARIFY Injection, please see Full Prescribing Information.
INDICATION
PYLARIFY (piflufolastat F 18) Injection is a radioactive diagnostic agent indicated for positron emission tomography (PET) of prostate-specific membrane antigen (PSMA) positive lesions in men with prostate cancer:
- with suspected metastasis who are candidates for initial definitive therapy.
- with suspected recurrence based on elevated serum prostate-specific antigen (PSA) level.
PYLARIFY (piflufolastat F 18) Injection is a radioactive diagnostic agent indicated for positron emission tomography (PET) of prostate-specific membrane antigen (PSMA) positive lesions in men with prostate cancer:
- with suspected metastasis who are candidates for initial definitive therapy.
- with suspected recurrence based on elevated serum prostate-specific antigen (PSA) level.
IMPORTANT SAFETY INFORMATION
Contraindications
None.
Warnings and Precautions
Risk of Image Misinterpretation
Imaging interpretation errors can occur with PYLARIFY imaging. A negative image does not rule out the presence of prostate cancer and a positive image does not confirm the presence of prostate cancer. The performance of PYLARIFY for imaging of patients with biochemical evidence of recurrence of prostate cancer seems to be affected by serum PSA levels. The performance of PYLARIFY for imaging of metastatic pelvic lymph nodes prior to initial definitive therapy seems to be affected by risk factors such as Gleason score and tumor stage. PYLARIFY uptake is not specific for prostate cancer and may occur with other types of cancer as well as non-malignant processes and in normal tissues. Clinical correlation, which may include histopathological evaluation of the suspected prostate cancer site, is recommended.
Hypersensitivity Reactions
Monitor patients for hypersensitivity reactions, particularly patients with a history of allergy to other drugs and foods. Reactions may be delayed. Always have trained staff and resuscitation equipment available.
Radiation Risks
Diagnostic radiopharmaceuticals, including PYLARIFY, expose patients to radiation. Radiation exposure is associated with a dose-dependent increased risk of cancer. Ensure safe handling and preparation procedures to protect patients and health care workers from unintentional radiation exposure. Advise patients to hydrate before and after administration and to void frequently after administration.
Adverse Reactions
The most frequently reported adverse reactions were headaches, dysgeusia and fatigue, occurring at rate of ≤2% during clinical studies with PYLARIFY. In addition, a delayed hypersensitivity reaction was reported in one patient (0.2%) with a history of allergic reactions.
Drug Interactions
Androgen deprivation therapy (ADT) and other therapies targeting the androgen pathway, such as androgen receptor antagonists, may result in changes in uptake of PYLARIFY in prostate cancer. The effect of these therapies on performance of PYLARIFY PET has not been established.
To report suspected adverse reactions for PYLARIFY, call 1-800-362-26681-800-362-2668 or contact FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
For important risk and use information about PYLARIFY Injection, please see Full Prescribing Information.
Contraindications
None.
Warnings and Precautions
Risk of Image Misinterpretation
Imaging interpretation errors can occur with PYLARIFY imaging. A negative image does not rule out the presence of prostate cancer and a positive image does not confirm the presence of prostate cancer. The performance of PYLARIFY for imaging of patients with biochemical evidence of recurrence of prostate cancer seems to be affected by serum PSA levels. The performance of PYLARIFY for imaging of metastatic pelvic lymph nodes prior to initial definitive therapy seems to be affected by risk factors such as Gleason score and tumor stage. PYLARIFY uptake is not specific for prostate cancer and may occur with other types of cancer as well as non-malignant processes and in normal tissues. Clinical correlation, which may include histopathological evaluation of the suspected prostate cancer site, is recommended.
Hypersensitivity Reactions
Monitor patients for hypersensitivity reactions, particularly patients with a history of allergy to other drugs and foods. Reactions may be delayed. Always have trained staff and resuscitation equipment available.
Radiation Risks
Diagnostic radiopharmaceuticals, including PYLARIFY, expose patients to radiation. Radiation exposure is associated with a dose-dependent increased risk of cancer. Ensure safe handling and preparation procedures to protect patients and health care workers from unintentional radiation exposure. Advise patients to hydrate before and after administration and to void frequently after administration.
Adverse Reactions
The most frequently reported adverse reactions were headaches, dysgeusia and fatigue, occurring at rate of ≤2% during clinical studies with PYLARIFY. In addition, a delayed hypersensitivity reaction was reported in one patient (0.2%) with a history of allergic reactions.
Drug Interactions
Androgen deprivation therapy (ADT) and other therapies targeting the androgen pathway, such as androgen receptor antagonists, may result in changes in uptake of PYLARIFY in prostate cancer. The effect of these therapies on performance of PYLARIFY PET has not been established.
To report suspected adverse reactions for PYLARIFY, call 1-800-362-26681-800-362-2668 or contact FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
For important risk and use information about PYLARIFY Injection, please see Full Prescribing Information.
REFERENCE
- Data on file. Bedford, MA; Progenics Pharmaceuticals, Inc; 2024.


