Why choose PYLARIFY

Clear diagnostic performance
PYLARIFY® delivered image clarity and accuracy with consistent reader interpretation across 2 robust clinical studies2,3
See clinical study results
Clear patient impact
See the number of patients who had a change in initial intended treatment after a PYLARIFY scan in the CONDOR trial
See CONDOR study results
Clear availability
Convenient and reliable supply via our extensive, multi-partner 18F distributor network, with leading support you can count on1
See the PYLARIFY site locatorGuiding a clear path forward
PYLARIFY has provided healthcare professionals and their patients with the clear information vital to making PCa treatment decisions.2
PYLARIFY can give your patients clarity


PYLARIFY can give your patients clarity
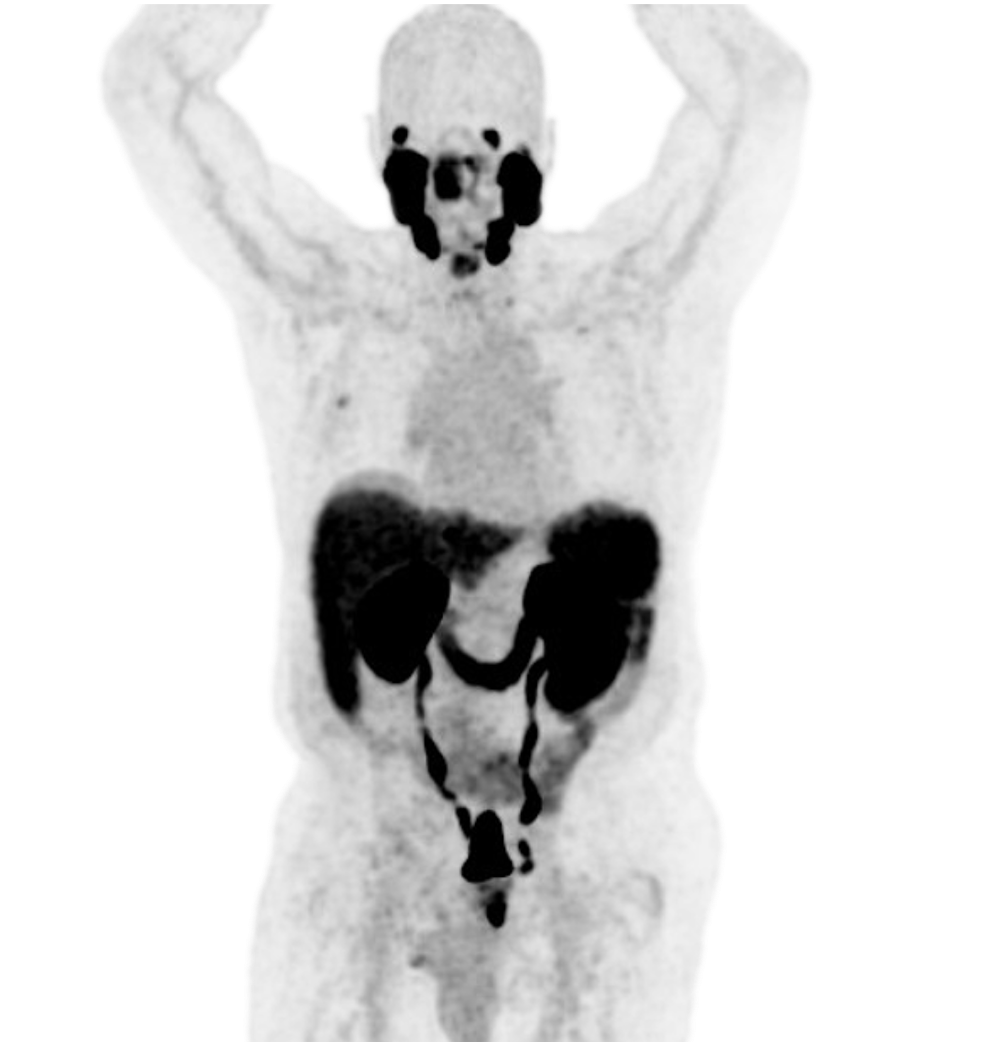
Discover clarity in the real world
Clear support from Lantheus
INDICATION
PYLARIFY (piflufolastat F 18) Injection is a radioactive diagnostic agent indicated for positron emission tomography (PET) of prostate-specific membrane antigen (PSMA) positive lesions in men with prostate cancer:
- with suspected metastasis who are candidates for initial definitive therapy.
- with suspected recurrence based on elevated serum prostate-specific antigen (PSA) level.
IMPORTANT SAFETY INFORMATION
Contraindications
None.
Warnings and Precautions
Risk of Image Misinterpretation
Imaging interpretation errors can occur with PYLARIFY imaging. A negative image does not rule out the presence of prostate cancer and a positive image does not confirm the presence of prostate cancer. The performance of PYLARIFY for imaging of patients with biochemical evidence of recurrence of prostate cancer seems to be affected by serum PSA levels. The performance of PYLARIFY for imaging of metastatic pelvic lymph nodes prior to initial definitive therapy seems to be affected by risk factors such as Gleason score and tumor stage. PYLARIFY uptake is not specific for prostate cancer and may occur with other types of cancer as well as non-malignant processes and in normal tissues. Clinical correlation, which may include histopathological evaluation of the suspected prostate cancer site, is recommended.
Hypersensitivity Reactions
Monitor patients for hypersensitivity reactions, particularly patients with a history of allergy to other drugs and foods. Reactions may be delayed. Always have trained staff and resuscitation equipment available.
Radiation Risks
Diagnostic radiopharmaceuticals, including PYLARIFY, expose patients to radiation. Radiation exposure is associated with a dose-dependent increased risk of cancer. Ensure safe handling and preparation procedures to protect patients and health care workers from unintentional radiation exposure. Advise patients to hydrate before and after administration and to void frequently after administration.
Adverse Reactions
The most frequently reported adverse reactions were headaches, dysgeusia and fatigue, occurring at rate of ≤2% during clinical studies with PYLARIFY. In addition, a delayed hypersensitivity reaction was reported in one patient (0.2%) with a history of allergic reactions.
Drug Interactions
Androgen deprivation therapy (ADT) and other therapies targeting the androgen pathway, such as androgen receptor antagonists, may result in changes in uptake of PYLARIFY in prostate cancer. The effect of these therapies on performance of PYLARIFY PET has not been established.
To report suspected adverse reactions for PYLARIFY, call 1-800-362-26681-800-362-2668 or contact FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
For important risk and use information about PYLARIFY Injection, please see Full Prescribing Information.
INDICATION
PYLARIFY (piflufolastat F 18) Injection is a radioactive diagnostic agent indicated for positron emission tomography (PET) of prostate-specific membrane antigen (PSMA) positive lesions in men with prostate cancer:
- with suspected metastasis who are candidates for initial definitive therapy.
- with suspected recurrence based on elevated serum prostate-specific antigen (PSA) level.
PYLARIFY (piflufolastat F 18) Injection is a radioactive diagnostic agent indicated for positron emission tomography (PET) of prostate-specific membrane antigen (PSMA) positive lesions in men with prostate cancer:
- with suspected metastasis who are candidates for initial definitive therapy.
- with suspected recurrence based on elevated serum prostate-specific antigen (PSA) level.
IMPORTANT SAFETY INFORMATION
Contraindications
None.
Warnings and Precautions
Risk of Image Misinterpretation
Imaging interpretation errors can occur with PYLARIFY imaging. A negative image does not rule out the presence of prostate cancer and a positive image does not confirm the presence of prostate cancer. The performance of PYLARIFY for imaging of patients with biochemical evidence of recurrence of prostate cancer seems to be affected by serum PSA levels. The performance of PYLARIFY for imaging of metastatic pelvic lymph nodes prior to initial definitive therapy seems to be affected by risk factors such as Gleason score and tumor stage. PYLARIFY uptake is not specific for prostate cancer and may occur with other types of cancer as well as non-malignant processes and in normal tissues. Clinical correlation, which may include histopathological evaluation of the suspected prostate cancer site, is recommended.
Hypersensitivity Reactions
Monitor patients for hypersensitivity reactions, particularly patients with a history of allergy to other drugs and foods. Reactions may be delayed. Always have trained staff and resuscitation equipment available.
Radiation Risks
Diagnostic radiopharmaceuticals, including PYLARIFY, expose patients to radiation. Radiation exposure is associated with a dose-dependent increased risk of cancer. Ensure safe handling and preparation procedures to protect patients and health care workers from unintentional radiation exposure. Advise patients to hydrate before and after administration and to void frequently after administration.
Adverse Reactions
The most frequently reported adverse reactions were headaches, dysgeusia and fatigue, occurring at rate of ≤2% during clinical studies with PYLARIFY. In addition, a delayed hypersensitivity reaction was reported in one patient (0.2%) with a history of allergic reactions.
Drug Interactions
Androgen deprivation therapy (ADT) and other therapies targeting the androgen pathway, such as androgen receptor antagonists, may result in changes in uptake of PYLARIFY in prostate cancer. The effect of these therapies on performance of PYLARIFY PET has not been established.
To report suspected adverse reactions for PYLARIFY, call 1-800-362-26681-800-362-2668 or contact FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
For important risk and use information about PYLARIFY Injection, please see Full Prescribing Information.
Contraindications
None.
Warnings and Precautions
Risk of Image Misinterpretation
Imaging interpretation errors can occur with PYLARIFY imaging. A negative image does not rule out the presence of prostate cancer and a positive image does not confirm the presence of prostate cancer. The performance of PYLARIFY for imaging of patients with biochemical evidence of recurrence of prostate cancer seems to be affected by serum PSA levels. The performance of PYLARIFY for imaging of metastatic pelvic lymph nodes prior to initial definitive therapy seems to be affected by risk factors such as Gleason score and tumor stage. PYLARIFY uptake is not specific for prostate cancer and may occur with other types of cancer as well as non-malignant processes and in normal tissues. Clinical correlation, which may include histopathological evaluation of the suspected prostate cancer site, is recommended.
Hypersensitivity Reactions
Monitor patients for hypersensitivity reactions, particularly patients with a history of allergy to other drugs and foods. Reactions may be delayed. Always have trained staff and resuscitation equipment available.
Radiation Risks
Diagnostic radiopharmaceuticals, including PYLARIFY, expose patients to radiation. Radiation exposure is associated with a dose-dependent increased risk of cancer. Ensure safe handling and preparation procedures to protect patients and health care workers from unintentional radiation exposure. Advise patients to hydrate before and after administration and to void frequently after administration.
Adverse Reactions
The most frequently reported adverse reactions were headaches, dysgeusia and fatigue, occurring at rate of ≤2% during clinical studies with PYLARIFY. In addition, a delayed hypersensitivity reaction was reported in one patient (0.2%) with a history of allergic reactions.
Drug Interactions
Androgen deprivation therapy (ADT) and other therapies targeting the androgen pathway, such as androgen receptor antagonists, may result in changes in uptake of PYLARIFY in prostate cancer. The effect of these therapies on performance of PYLARIFY PET has not been established.
To report suspected adverse reactions for PYLARIFY, call 1-800-362-26681-800-362-2668 or contact FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
For important risk and use information about PYLARIFY Injection, please see Full Prescribing Information.
References
- Data on file. Bedford, MA: Progenics Pharmaceuticals, Inc.; 2025.
- Morris MJ, Rowe SP, Gorin MA, et al. Diagnostic performance of 18F-DCFPyL-PET/CT in men with biochemically recurrent prostate cancer: results from the CONDOR phase III, multicenter study. Clin Cancer Res. 2021;27(13):3674-3682.
- Pienta KJ, Gorin MA, Rowe SP, et al. A phase 2/3 prospective multicenter study of the diagnostic accuracy of prostate specific membrane antigen PET/CT with 18F-DCFPyL in prostate cancer patients (OSPREY). J Urol. 2021;206(1):52-61.

